Quick Summary
A comprehensive guide to Degenerative Cervical Myelopathy (DCM). From the subtle 'Finger Escape' sign to the choice between ACDF and Laminoplasty.
Cervical Myelopathy: The Silent Progression
Degenerative Cervical Myelopathy (DCM) is recognized as the most common cause of spinal cord dysfunction in adults worldwide. Yet, it remains one of the most frequently missed diagnoses in primary care and even in busy orthopaedic outpatient clinics. Because the condition is often entirely painless in its early stages, it creeps up on patients. They present with vague complaints of "clumsiness," "old age balance issues," or "stiff legs," only to be definitively diagnosed when they have already suffered irreversible spinal cord damage.
For the orthopaedic surgeon—and particularly for those in orthopaedic surgery training preparing for fellowship exams—detecting DCM early and understanding the nuances of its management is a critical, foundational skill. Failing to identify myelopathy before it progresses can lead to devastating consequences for the patient's independence and quality of life.
This comprehensive guide outlines the underlying pathophysiology, the subtle and overt clinical signs, the pivotal role of advanced imaging, and the nuanced surgical decision-making algorithms that define modern spine surgery management.
The Pathophysiology of Cord Compression
The spinal cord in the cervical spine operates in a remarkably tight physiological space. The normal anterior-posterior (AP) canal diameter is roughly 17-18mm. Myelopathy typically begins to manifest when this critical space narrows to less than 13mm, a state that can be congenital, acquired, or a combination of both.
Understanding the pathophysiology is paramount for fellowship exam preparation, as examiners frequently test the distinction between static and dynamic factors.
The Two Primary Mechanisms of Injury
- Static Compression: This involves direct, unrelenting pressure on the spinal cord. It is most commonly caused by anterior structures such as spondylotic osteophytes (hard disc complexes), extruded disc herniations (soft discs), or Ossification of the Posterior Longitudinal Ligament (OPLL). Posteriorly, facet hypertrophy and thickened ligamentum flavum contribute. This sustained pressure leads to localized ischemia, venous congestion, and ultimately, apoptosis of oligodendrocytes and neurons.
- Dynamic Compression: The cervical spine is a highly mobile segment, and physiological movements can exacerbate stenosis. In cervical extension, the spinal canal shortens and the ligamentum flavum buckles inward (often referred to as "shingling"), severely compressing the posterior columns. In cervical flexion, the spinal cord is stretched and draped over anterior spondylotic bars or osteophytes, increasing longitudinal tension and anterior cord pressure. This repetitive micro-trauma leads to demyelination and irreversible axonal damage over time.
A classic radiographic measurement tested in surgical education. It is the ratio of the AP diameter of the spinal canal to the AP diameter of the vertebral body on a lateral C-spine X-ray. A ratio of < 0.8 indicates congenital cervical stenosis, a significant risk factor for developing early-onset DCM or sustaining central cord syndrome following minor trauma.
Visual Element: Cross-section diagram showing the cord flattened into a "banana shape" or "boomerang" by anterior osteophytes and posterior ligamentum flavum buckling, obliterating the subarachnoid space and CSF buffer.
Clinical Presentation: Unmasking the "Myelopathy Hand"
Pain is an incredibly unreliable indicator of DCM. Radicular arm pain or axial neck pain may be present, but pure myelopathy is often completely painless. The astute clinician must look for deficits in function, dexterity, and proprioception.
Subjective Complaints: Listening to the Patient
When taking a history during a fellowship exam OSCE or in clinic, pay close attention to the specific ways patients describe their functional loss:
- Upper Extremities (Dexterity Loss): "I can't button my dress shirt anymore." "I keep dropping my coffee cup." "My handwriting has become slow and illegible." "I have trouble picking up coins from the table."
- Lower Extremities (Gait and Proprioception): "I feel like I'm walking on cotton wool or a bouncy castle." "I'm incredibly unsteady when I get up to go to the bathroom in the dark." "My legs feel heavy and stiff."
- Sphincter Dysfunction: Urinary urgency, frequency, or hesitancy. Overt incontinence is typically a very late and grave sign.
Objective Physical Examination: Upper Motor Neuron Signs
A meticulous neurological examination is the cornerstone of diagnosing DCM. The presence of Upper Motor Neuron (UMN) signs below the level of compression, often mixed with Lower Motor Neuron (LMN) signs at the specific level of root compression (myeloradiculopathy), is the hallmark of the disease.
- The Finger Escape Sign (Wartenberg's Sign of Myelopathy): Ask the patient to hold their hands out with fingers fully extended and adducted. Watch the ulnar digits. The little finger (and sometimes the ring finger) will spontaneously drift into abduction and flexion. This occurs due to subtle weakness of the intrinsic hand muscles combined with mild spasticity. It mimics an ulnar nerve palsy, but notably, there is no associated sensory loss in the ulnar distribution.
- Grip and Release Test (10-Second Test): Ask the patient to make a tight fist and fully extend their fingers as fast as possible. A normal adult can perform this complete cycle >20 times in 10 seconds. Myelopathic patients will be noticeably slow, clumsy, and unable to fully extend their digits (often managing fewer than 10-15 cycles).
- Hoffmann's Sign: Stabilize the patient's middle phalanx of the 3rd digit and briskly flick the distal phalanx downward. A positive sign is the reflexive flexion of the thumb IP joint and/or the index finger tip. This indicates cervical cord irritability/hyperreflexia.
- Inverted Radial Reflex: Tapping the brachioradialis tendon at the distal radius normally elicits elbow flexion. In a positive inverted reflex (due to a C5/C6 level cord compression), the reflex arc is altered, and you will observe spontaneous finger flexion instead of, or in addition to, wrist/elbow movement.
- Lhermitte's Sign: An electrical shock-like sensation radiating down the spine or into the limbs upon active or passive cervical flexion. This indicates tension on an irritable posterior column.
- Lower Limb Hyperreflexia & Clonus: Markedly brisk patellar and Achilles reflexes. Sustained ankle clonus (>3 beats) is a definitive UMN sign.
- Tandem Gait Test: Ask the patient to walk heel-to-toe in a straight line. Patients with proprioceptive loss and spasticity will inevitably lose their balance and require a wide-based stance to compensate.
Diagnostic Pitfall: Carpal Tunnel Syndrome
DCM is frequently misdiagnosed as bilateral Carpal Tunnel Syndrome (CTS) due to complaints of hand clumsiness and subjective numbness. Always check lower limb reflexes in a patient presenting with "CTS". Brisk knees or ankles in a patient with clumsy hands strongly suggest the pathology is in the cervical cord, not the wrist.
Standardized Classification Systems
In surgical education and clinical practice, we don't just say a patient has "myelopathy." We must quantify the severity to guide treatment algorithms and track progression. Familiarity with these scores is mandatory for fellowship exam preparation.
1. The Nurick Grade
The Nurick system is primarily focused on ambulatory status and gait impairment. It is simple and easy to use in a busy clinic.
- Grade 0: Root signs and symptoms only; no evidence of spinal cord disease.
- Grade 1: Signs of spinal cord disease (e.g., hyperreflexia) but no difficulty in walking.
- Grade 2: Slight difficulty in walking, but does not prevent full-time employment.
- Grade 3: Difficulty walking that prevents full-time employment or the ability to do all daily chores, but walking unassisted.
- Grade 4: Able to walk only with the assistance of a cane, walker, or another person.
- Grade 5: Chair-bound or bedridden.
2. Modified Japanese Orthopaedic Association (mJOA) Score
The mJOA is the most widely validated tool for assessing DCM severity. It is a 18-point scale (often cited as 17 or 18 depending on the exact modification used, but commonly out of 18 in modern literature) assessing four domains: Upper Limb Motor function (5 points), Lower Limb Motor function (7 points), Upper Limb Sensation (3 points), and Sphincter function (3 points).
- Mild Myelopathy: mJOA 15-17
- Moderate Myelopathy: mJOA 12-14
- Severe Myelopathy: mJOA < 12
Exam Tip: mJOA vs. JOA
The original JOA score included an assessment of chopstick use, which is culturally specific. The modified JOA (mJOA) replaces this with a spoon/fork assessment or general buttoning capability, making it globally applicable. Always specify mJOA in your exam answers.
The Crucial Role of Imaging
While clinical examination raises the suspicion, advanced imaging confirms the anatomical level and the degree of cord injury.
Magnetic Resonance Imaging (MRI): The Gold Standard
MRI is non-negotiable for evaluating suspected DCM.
- T2-Weighted Sequences: Look for high-signal intensity (white) within the spinal cord itself at the level of maximum compression.
- Faint, fuzzy, or broad high signal typically represents cord edema and reversible ischemia.
- Bright, sharp, well-demarcated high signal (the "Snake Eyes" or "Owl Eyes" appearance on axial views) indicates cystic necrosis and myelomalacia, which is often irreversible.
- T1-Weighted Sequences: Look for low-signal intensity (black) within the cord. A T1 "black hole" correlates with focal cavitation, severe neuronal loss, and a fundamentally poor prognosis for neurological recovery even after perfect surgical decompression.
Plain Radiographs and CT
- Dynamic Flexion/Extension X-Rays: Essential to rule out dynamic instability (e.g., degenerative spondylolisthesis) which would mandate a fusion procedure rather than a motion-preserving decompression.
- CT Scan: Invaluable for assessing bony anatomy, planning screw trajectories, and specifically for identifying OPLL (which is often difficult to distinguish from the posterior longitudinal ligament on MRI alone).
Management Algorithms: To Cut or Not to Cut?
The natural history of DCM is generally characterized by a stepwise deterioration. Patients may remain stable for months or years, followed by an abrupt, unpredictable decline in function. It rarely, if ever, improves spontaneously.
The primary goal of surgical management is to arrest the progression of the disease. Any neurological recovery is considered a bonus, not a guarantee. This must be clearly communicated to the patient during informed consent.
- Moderate to Severe DCM (mJOA < 14): Surgical decompression is strongly recommended. The risks of non-operative management (irreversible paralysis) far outweigh the risks of surgery.
- Mild DCM (mJOA 15-17): Management here is more nuanced and highly tested in orthopaedic surgery training. Options include:
- Close clinical surveillance: Frequent follow-ups (every 3-6 months) with repeat mJOA scoring. If any neurological deterioration occurs -> prompt surgery.
- Proactive Surgery: Recommended if the patient has very tight stenosis, high-risk features (T2 signal change), or lifestyle factors that make close follow-up difficult.
Surgical Approaches: The Decision-Making Matrix
The overarching principle is simple: increase the space for the spinal cord. The decision of how to achieve this—Anterior vs. Posterior vs. Combined—depends on sagittal alignment, the location of the pathology, and the number of levels involved.
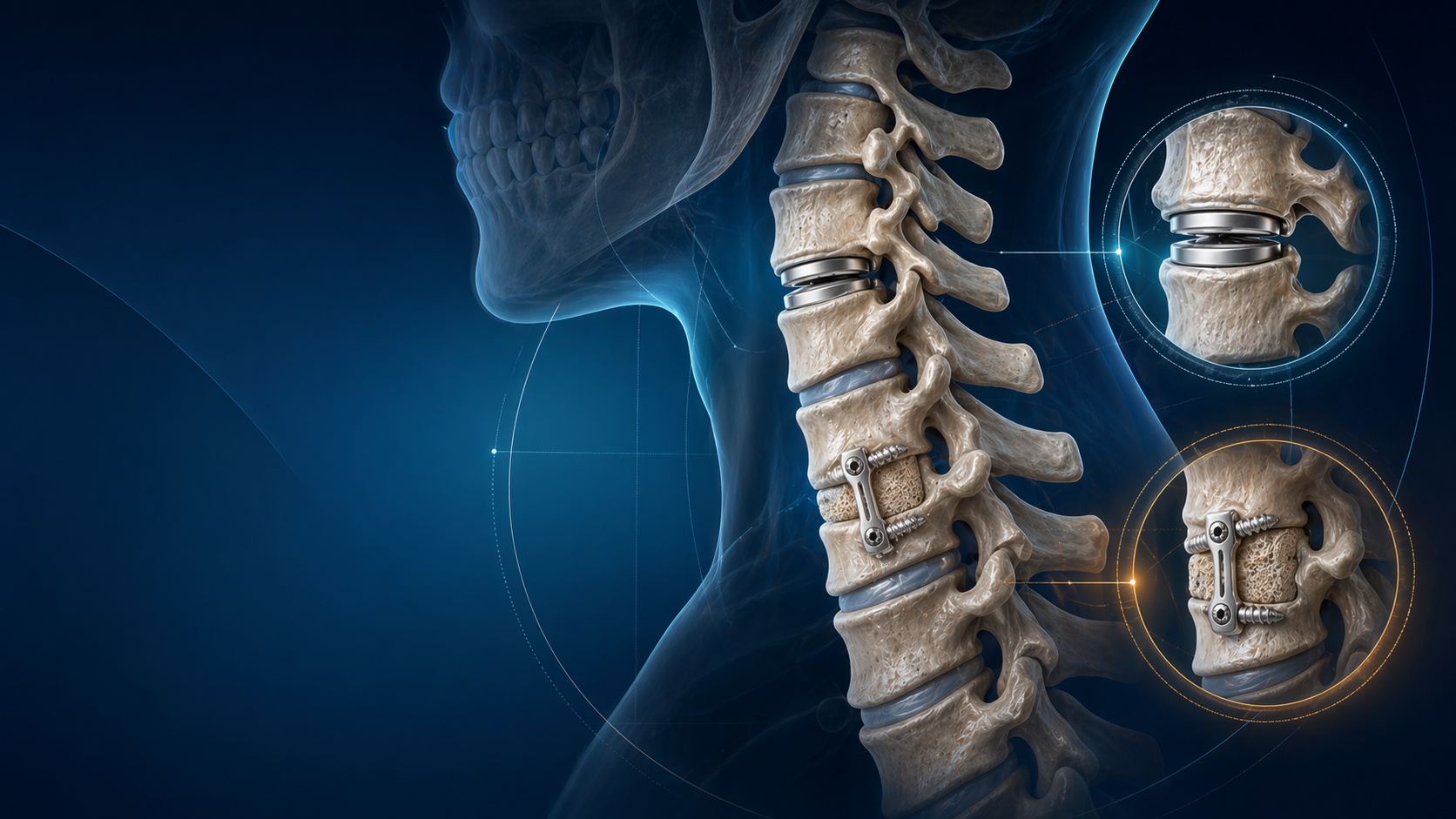
1. Anterior Approaches (ACDF / Cervical Corpectomy)
- Indications:
- Pathology limited to 1-3 levels.
- Predominantly anterior compression (large soft disc herniations, localized osteophytes).
- Kyphotic cervical alignment. (Anterior surgery allows the surgeon to place lordotic cages, actively correcting the sagittal deformity).
- Technique: Anterior Cervical Discectomy and Fusion (ACDF) involves removing the intervertebral disc and posterior osteophytes back to the posterior longitudinal ligament, then placing an interbody cage and an anterior plate.
- Pros: Direct removal of the offending pathology. Excellent ability to restore cervical lordosis. Very high fusion rates. Indirect decompression of the foramina.
- Cons: Risk of postoperative dysphagia (swallowing difficulty) and dysphonia (recurrent laryngeal nerve neuropraxia). Risk of Adjacent Segment Disease (ASD) over time, estimated at roughly 2-3% per year.
2. Posterior Approaches: Cervical Laminoplasty
- Indications:
- Multi-level disease (typically >3 levels).
- Congenital cervical stenosis (narrow canal from C3 to C7).
- Extensive OPLL where an anterior approach carries a prohibitive risk of dural tear.
- Crucially: The patient MUST have preserved cervical lordosis (or at least neutral alignment).
- Technique: The lamina is hinged open ("Open Door" Hirabayashi technique or "French Door" Kurokawa technique) using mini-plates or bone graft to prop the canal open. This expands the space available for the cord without committing to a rigid fusion.
- Pros: Preserves some cervical range of motion. Avoids the complications associated with bone grafting and pseudoarthrosis. Less risk of adjacent segment disease.
- Cons: Can lead to persistent posterior neck pain. Carries a significant risk of postoperative C5 palsy.
3. Posterior Approaches: Laminectomy and Fusion
- Indications:
- Multi-level disease combined with dynamic instability.
- Multi-level disease in a patient with a flexible kyphotic deformity (where the fusion constructs the spine in a corrected, lordotic position).
- Previous failed anterior surgery.
- Technique: Complete removal of the lamina and spinous processes to definitively unroof the cord, followed by stabilization using lateral mass or pedicle screws and rods.
- Pros: Extremely robust and definitive decompression. Rigid stabilization prevents dynamic micro-trauma.
- Cons: Complete loss of motion at the fused segments. Higher surgical morbidity (blood loss, infection risk) compared to laminoplasty. Risk of adjacent segment disease.
Never perform an isolated laminectomy or laminoplasty in a fixed kyphotic spine. If the spine is kyphotic, the spinal cord is draped tightly over the anterior vertebral bodies—like a bowstring over a bow. Removing the posterior bony arch will not decompress the cord because the cord will not "drift back" away from the anterior pathology. In kyphotic cases, you must address the deformity anteriorly (to restore lordosis) or via a combined anterior-posterior approach.
Specific Surgical Complications: Focus on C5 Palsy
While generic surgical complications (infection, bleeding, dural tears) exist, C5 palsy is uniquely relevant to cervical decompression for myelopathy and is a favorite topic in fellowship exams.
- The Clinical Picture: A patient wakes up from a successful posterior (or anterior) multilevel decompression with intact neural function. Between 2 to 5 days post-operatively, they develop profound weakness in their deltoid and biceps (inability to abduct the shoulder or flex the elbow). Sensation is usually preserved, and there is no radicular pain.
- Pathophysiology Theories:
- The Tethering Effect: As the spinal cord drifts backward into the newly expanded canal, the short, horizontal C5 nerve roots are put under immense traction and stretch.
- Reperfusion Injury: Sudden restoration of blood flow to a chronically ischemic segment of the cord leads to oxidative stress and localized edema affecting the anterior horn cells.
- Management and Prognosis: First, urgently repeat an MRI to rule out an epidural hematoma or iatrogenic foraminal stenosis. If the scan is clear, management is conservative (physical therapy to prevent shoulder contractures).
- Outcome: The majority of patients will see spontaneous resolution, but it demands patience. Recovery can take anywhere from 3 to 12 months, and severe cases may have permanent residual weakness.
Conclusion
Degenerative Cervical Myelopathy is a diagnosis of high clinical consequence that must not be missed by the orthopaedic surgeon. The window of opportunity to intervene and preserve neurological function is often distressingly narrow.
When evaluating an aging patient who complains of "clumsy hands," dropping objects, and frequent trips or falls, the differential diagnosis must aggressively prioritize the cervical spinal cord. Look beyond the wrist; assess their tandem gait, check for hyperreflexia, and flick that middle finger. Remember: suspecting the diagnosis is the hardest part. Once confirmed, a structured approach to imaging and surgical planning can halt the silent progression of this devastating disease.
References
- Fehlings, M. G., Tetreault, L. A., Riew, K. D., et al. (2017). "A Clinical Practice Guideline for the Management of Patients With Degenerative Cervical Myelopathy: Recommendations for Patients With Mild, Moderate, and Severe Disease and Nonmyelopathic Patients With Evidence of Cord Compression." Global Spine Journal, 7(3_suppl), 70S-83S. This landmark paper provides the current evidence-based algorithms for treating varying severities of DCM.
- Nurick, S. (1972). "The natural history and the results of surgical treatment of the spinal cord disorder associated with cervical spondylosis." Brain, 95(1), 101-108. The original description of the classic Nurick grading system.
- Kato, S., Nouri, A., Wu, D., Nori, S., Tetreault, L., & Fehlings, M. G. (2016). "Comparison of Anterior and Posterior Approaches for the Treatment of Multilevel Cervical Spondylotic Myelopathy." Global Spine Journal, 6(3), 296-302. Excellent review of decision-making for complex multi-level disease.
- Emery, S. E. (2001). "Cervical Spondylotic Myelopathy: Diagnosis and Treatment." Journal of the American Academy of Orthopaedic Surgeons, 9(6), 376-388. A foundational orthopaedic text detailing the physical examination and surgical rationale.
Found this helpful?
Share it with your colleagues